(2008) The cytoplasmic loops of subunit a of Escherichia coli ATP synthase may participate in the proton translocating mechanism. Moore, Kyle J Angevine, Christine M Vincent, Owen D et al. Steed, P Ryan Fillingame, Robert H (2009) Aqueous accessibility to the transmembrane regions of subunit c of the Escherichia coli F1F0 ATP synthase. Protein Sci 21:279-88ĭong, Hui Fillingame, Robert H (2010) Chemical reactivities of cysteine substitutions in subunit a of ATP synthase define residues gating H+ transport from each side of the membrane. (2012) Cell-free synthesis of membrane subunits of ATP synthase in phospholipid bicelles: NMR shows subunit a fold similar to the protein in the cell membrane. Uhlemann, Eva-Maria E Pierson, Hannah E Fillingame, Robert H et al. (2013) Interactions between subunits a and b in the rotary ATP synthase as determined by cross-linking. J Biol Chem 288:25535-41ĭeLeon-Rangel, Jessica Ishmukhametov, Robert R Jiang, Warren et al. Moore, Kyle J Fillingame, Robert H (2013) Obstruction of transmembrane helical movements in subunit a blocks proton pumping by F1Fo ATP synthase. Steed, P Ryan Fillingame, Robert H (2014) Residues in the polar loop of subunit c in Escherichia coli ATP synthase function in gating proton transport to the cytoplasm. Steed, P Ryan Kraft, Kaitlin A Fillingame, Robert H (2014) Interacting cytoplasmic loops of subunits a and c of Escherichia coli F1F0 ATP synthase gate H+ transport to the cytoplasm. The principles by which this enzyme works may provide fundamental insights into other transport problems in biology and medicine.įillingame, Robert H Steed, P Ryan (2014) Half channels mediating H(+) transport and the mechanism of gating in the Fo sector of Escherichia coli F1Fo ATP synthase. Closely related enzymes are responsible for vesicular acidification in human cells, and work by a similar rotary mechanism. Abnormalities in the enzyme lead to human disease. The ATP synthase is central to cellular function-it makes the ATP. Ultimately, we hope to define an atomic resolution structure that can be used in mechanistic studies. Initially, the global fold of the purified protein in solution will be compared to that in the membrane using spin-labeled protein to establish appropriate solution conditions. Simultaneously, we will attempt to determine the solution structure of purified subunit a by NMR. 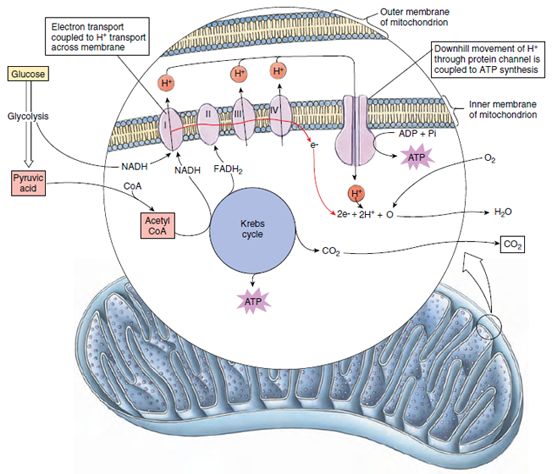
Aqueous access pathways in subunit a mediating H+ transport from membrane surfaces to the H+ binding site in subunit c will be defined, and the mechanism of gating H+ access to the two sides of the membrane probed. The global fold and packing of subunit a in native Escherichia coli membranes will be determined by cross link analysis. This proposal focuses on the structure of subunit a, with the ultimate goal of defining its role in coupling H+ transport to c-ring rotation. The concerted rotation of helices at the subunit a-c interface is proposed to mechanically drive the stepwise movement of the c-ring. Biochemical evidence indicates that one of the helices of subunit c, which resides at the interface with subunit a, rotates between two different conformations. The structure of subunit c was solved by solution NMR and the c-ring has been modeled. The mechanism of coupling H+ transport and c-ring rotation is poorly understood. A stator complex of F0 subunits a and b and F1 subunit delta extends from the membrane to the top of the F1 molecule and holds alpha-3-beta-3 fixed, relative to the membrane, allowing the c-gamma complex to rotate within. H+ transport through transmembrane F0 drives rotation of an oligomeric ring of c subunits connected with gamma, and results in ATP synthesis in catalytic sites at the alpha-beta interface. Rotation of subunit gamma within the core of the alpha-3-beta-3 hexamer of F1 drives ATP synthesis by a unique rotary catalytic mechanism. The H+-transporting F1F0 ATP synthases of oxidative phosphorylation in mitochondria and bacteria are very similar. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
